For the first time, physicists have succeeded in creating a strange, fragile type of structure in the laboratory known as the trilobite Rydberg molecule.
Building and observing these strange atomic structures has given scientists new insights into the quantum activity of electrons as they propagate near atoms.
Since their chemical bonds are unlike any other (as we know them), the results open up possibilities for developing better theoretical models of molecules and understanding their dynamics.
Rydberg molecules are created from a type of atom known as a Rydberg atom. In a regular atom, you have a nucleus, surrounded by a small swarm of electrons. If you add a little energy to the atom, the swarm of electrons swells up a little, making the atom a little larger and more flexible.
A Rydberg atom is what you get when you add a lot of energy under conditions that allow it to retain its electrons. It bulges very large, for an atom, several microns across, and the electrons are bound as loosely as possible without flying off.
Because they are incoherent, Rydberg atoms behave in an exaggerated manner, making them… Useful for conducting experiments.
Molecules are arrangements of atoms that are clustered together in some way, such as by shared electrons or perhaps by opposite charges. If you use a Rydberg atom, you get a Rydberg molecule, but the way the atoms stick together They can be very different from bonds Which joins more conventional molecules.
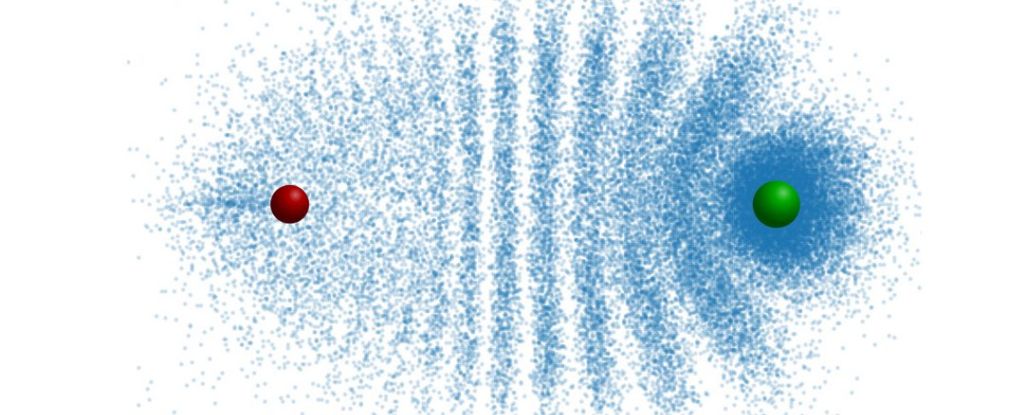
They can look very different, with electron distribution patterns that can resemble, for example, a Trilobitesor a butterfly.
Led by physicist Max Althon of the University of Kaiserslautern-Landau, a team of scientists in the Herwig Ott laboratory has, for the first time, created pure trilobite Rydberg molecules.
They started with rubidium atoms, supercooled to a temperature of just 0.0001 degrees above absolute zero. They then used lasers to excite some of the atoms into Rydberg states.
“In this process, the outermost electron is in each case brought into distant orbits around the atomic body,” Ott says. “The radius of the electron orbit can be more than one micrometer, making the electron cloud larger than a small bacterium.”
A Rydberg molecule can be created by bringing a ground-state atom—which has not been excited to the Rydberg state—into the swollen electron swarm of a Rydberg atom, where the two atoms are held together not by standard chemical bonds, but by strange quantum gravity. .
“It is the quantum mechanical scattering of the Rydberg electron from the ground state atom, which links the two together.” Alton explains.
“Imagine an electron rapidly orbiting the nucleus. On each round trip, it collides with a ground-state atom. Contrary to our intuition, quantum mechanics teaches us that these collisions lead to an effective attraction between the electron and the ground-state atom.”
Due to repeated collisions, the electrons are distributed in an interference pattern similar to the segmented shield of a trilobite.
It has other wonderful and strange properties as well. The molecular bond length is approximately the same size as the Rydberg orbital, which is very large by atomic scales. The force of attraction between the electron and the ground state atom is also very high.
This means that the Rydberg molecules have a higher Electric dipole moment than any other molecule; That is, the separation of positive and negative electrical charges, also known as polarity.
The trilobite Rydberg molecules observed by Alton and colleagues have an electric dipole moment of more than 1,700 Debbies, which is very high. For water molecules, this measurement is less than 2 Debye.
The ability to not only create pure Rydberg trilobites, but explore them, gives physicists a new tool to test and understand the quantum world.
It also has potential applications for quantum information processing. The researchers say it could be applied more widely to study these foreign molecules across different species.
“In conclusion, we have measured two vibrational chains of pure Rydberg trilobite molecules by using three-photon optical coherence,” they write. “In this way, it should be possible to create trilobite particles in any element with a negative wave dispersion length s.”
The research was published in Nature Communications.

“Extreme travel lover. Bacon fanatic. Troublemaker. Introvert. Passionate music fanatic.”





More Stories
Who is the band Gojira that will perform at the Olympics opening ceremony?
SpaceX Moves Crew Dragon Spacecraft to West Coast After Multiple Space Debris Incidents
Stathis Karapanos – Hindemith Review: Complete Works for Flute